Get Matched with Mesothelioma Clinical Trials
Our clinical trial support team helps patients:
- Identify active mesothelioma clinical trials in the U.S.
- Match with Phase 1, Phase 2, Phase 2/3, and Phase 3 studies
- Navigate eligibility requirements
- Coordinate appointments with leading cancer centers
Call now or request a free consultation to connect with a nurse who can guide you.
Vivace’s VT3989 for Mesothelioma
Table of Contents
- 1 What are Mesothelioma Clinical Trials?
- 2 Understanding Clinical Trial Phases
- 3 Latest Phase 1 Mesothelioma Clinical Trials
- 4 Latest Phase 2/3 and Phase 3 Mesothelioma Clinical Trials
- 5 Who Qualifies for a Mesothelioma Clinical Trial?
- 6 Costs and Insurance Considerations
- 7 Questions to Ask Before Joining a Trial
- 8 Talk to a Nurse About Mesothelioma Clinical Trials
- 9 Talk to a mesothelioma nurse specialist
Vivace’s VT3989 is an investigational therapy targeting the Hippo signaling pathway, which is disrupted in roughly 60% of mesothelioma cases. This drug works as a TEAD palmitoylation inhibitor, interfering with the activity of TEAD proteins that normally partner with YAP and TAZ to drive tumor growth when the Hippo pathway is turned off. By blocking this interaction, VT3989 aims to limit cancer cell proliferation and survival. It is currently being studied in patients with advanced, treatment-resistant solid tumors, including mesothelioma. Early research suggests that inhibiting the YAP–TEAD pathway may also enhance the effectiveness of immunotherapy by improving T cell access to tumors, although more data are needed to determine which patients are most likely to benefit. Our nurse navigator services are free of charge and can be used by our clients and non-clients alike. Contact our nurse to find out if you’re eligible for VT3989 trial.
Checkmate 743 for Mesothelioma
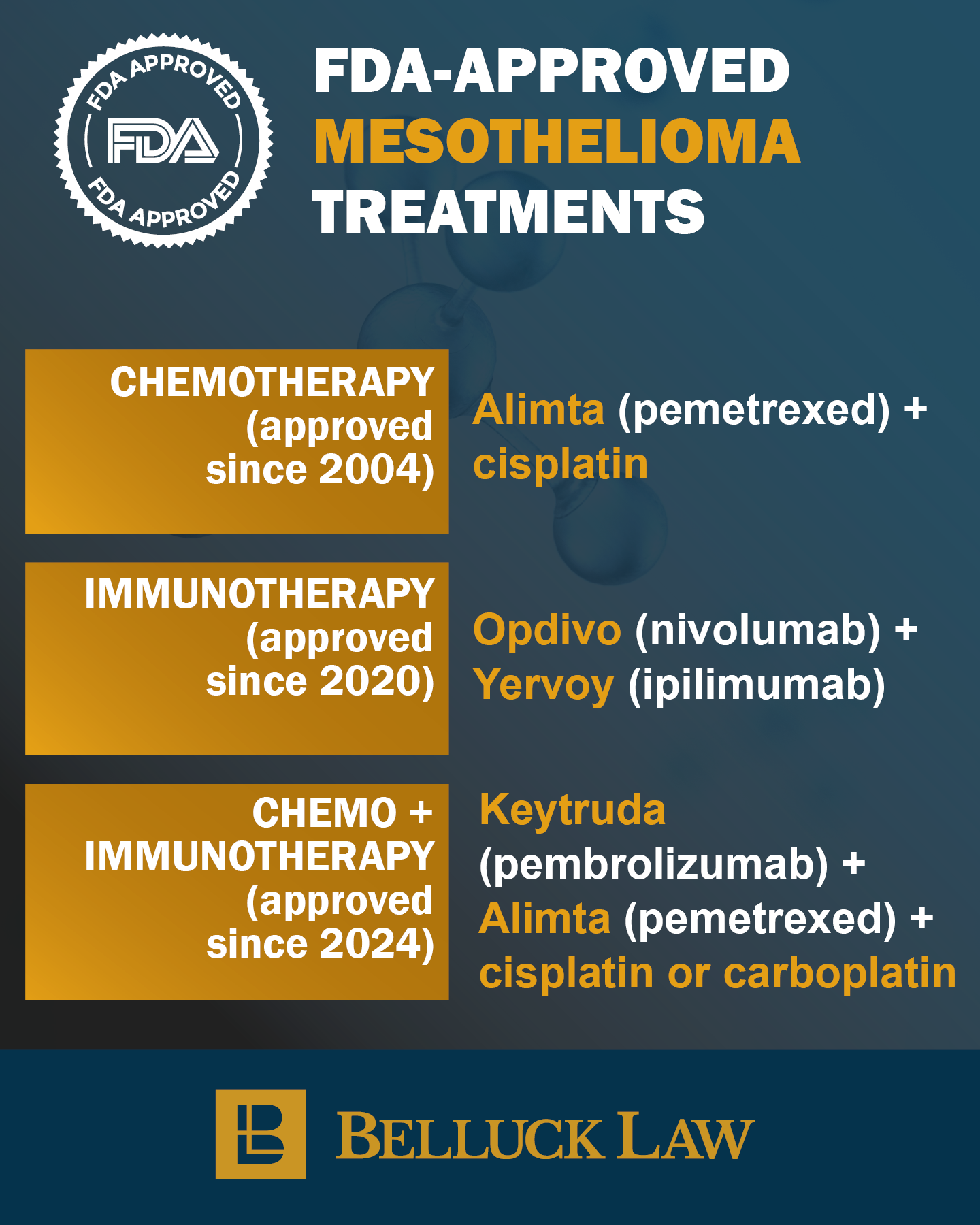
The combination of Opdivo and Yervoy represents a major advance in the first-line treatment of unresectable malignant pleural mesothelioma, working by activating the immune system through dual checkpoint inhibition targeting PD-1 and CTLA-4 pathways. Its approval by the U.S. Food and Drug Administration was based on the pivotal Phase 3 CheckMate 743 trial, which enrolled 605 previously untreated patients and demonstrated a statistically significant survival benefit over standard platinum-based chemotherapy. Patients receiving the immunotherapy combination achieved a median overall survival of 18.1 months compared to 14.1 months with chemotherapy, translating to a 26% reduction in the risk of death (HR 0.74, p=0.002). Longer-term follow-up has shown durable benefits, with higher two-year survival rates and sustained improvements in overall survival even out to five years in some patients. Opdivo plus Yervoy thus established a new standard of care, offering a chemotherapy-free option that can produce longer-lasting responses, although not all patients respond and ongoing research is focused on identifying predictive biomarkers. Our nurse navigator services are free of charge and can be used by our clients and non-clients alike. Contact our nurse to find out if you’re eligible for the Checkmate 743 trial.
eVOLVE-Meso
Volrustomig (MEDI5752) is an emerging next-generation immunotherapy being studied in mesothelioma through the global Phase 3 eVOLVE-Meso trial, which is evaluating its use in combination with standard chemotherapy for patients with unresectable pleural disease. Unlike earlier checkpoint inhibitors that target a single immune pathway, volrustomig is designed as a bispecific antibody that simultaneously blocks two immune checkpoints, with the goal of producing a more potent and coordinated anti-tumor immune response. In this randomized study that will ultimately enroll 600 patients, the combination of volrustomig plus carboplatin and pemetrexed is being compared against current standards of care, including chemotherapy alone or the nivolumab and ipilimumab (known as Opdivo and Yervoy) immunotherapy regimen. The trial, which began enrollment in 2023, is intended to determine whether this dual-targeting approach can improve survival and overall outcomes in the first-line setting, while also assessing safety and quality of life. Our nurse navigator services are free of charge and can be used by our clients and non-clients alike. Contact our nurse to find out if you’re eligible for VT3989 trial.
What are Mesothelioma Clinical Trials?
According to the National Cancer Institute, clinical trials are the final stage of research where new treatments are tested in patients. These studies evaluate whether emerging therapies are safe, effective, and better than current standards of care.
Mesothelioma clinical trials may include:
- New immunotherapy or targeted drug treatments
- Advanced chemotherapy combinations
- Surgical innovations
- Gene therapy and experimental approaches
- Comparisons between standard treatments and new therapies
Many current Phase 2/3 and Phase 3 mesothelioma clinical trials in the U.S. are focused on immunotherapy combinations and personalized treatment strategies.
Understanding Clinical Trial Phases
The Food and Drug Administration oversees the clinical trial process:
- Phase 1: Focus on safety and dosage
- Phase 2: Evaluates effectiveness in mesothelioma patients
- Phase 2/3: Combines effectiveness and expanded safety analysis
- Phase 3: Large-scale comparison to standard treatments
- Phase 4: Ongoing monitoring after approval
Most patients searching for mesothelioma clinical trials are eligible for Phase 2, Phase 2/3, or Phase 3 studies.
Latest Phase 1 Mesothelioma Clinical Trials
Most Phase 1 mesothelioma trials are exploring new approaches, including:
Next-Generation Immunotherapy
- Novel checkpoint inhibitors beyond standard PD-1/PD-L1 drugs
- Bispecific antibodies that target two immune pathways at once
- Engineered immune cell therapies (like CAR-T adapted for mesothelioma)
Focus: Can this safely activate the immune system against mesothelioma?
Gene and Cell-Based Therapies
- Viral gene therapies (similar to interferon-based delivery systems)
- Modified immune cells designed to recognize mesothelioma tumors
Focus: Is this safe, and does it reach and affect tumor cells?
Targeted Molecular Therapies
- Drugs aimed at specific mutations or tumor pathways
- RNA-based or antisense therapies
Focus: What dose works without unacceptable side effects?
Latest Phase 2/3 and Phase 3 Mesothelioma Clinical Trials
Recent data from ClinicalTrials.gov shows ongoing and newly posted trials across the country, including:
- Phase 2/3 trials evaluating combination immunotherapies for pleural mesothelioma
- Phase 3 trials comparing immunotherapy drugs to traditional chemotherapy
- Studies testing checkpoint inhibitors alongside surgery or radiation
- Trials exploring biomarker-driven treatments to match therapies to patient profiles
These advanced-stage trials are critical because they involve larger patient groups and are often the final step before potential FDA approval.
Who Qualifies for a Mesothelioma Clinical Trial?
Eligibility depends on several factors:
- Type and stage of mesothelioma
- Previous treatments received
- Overall health and medical history
- Specific trial criteria
Your doctor can help determine eligibility, but navigating options can be overwhelming.
Our nurse can review your diagnosis, match you with active trials, and help coordinate next steps.
Costs and Insurance Considerations
Most clinical trials are funded by government agencies, research institutions, or pharmaceutical companies. This often means:
- The trial treatment itself is covered
- Routine care (doctor visits, scans) may still go through insurance
- Travel and lodging may or may not be reimbursed
Always confirm coverage details with your provider before enrolling.
Questions to Ask Before Joining a Trial
Before enrolling in a mesothelioma clinical trial, consider asking:
- What treatment is being tested?
- What are the potential risks and benefits?
- How often are visits required?
- Will I receive standard treatment or a new therapy?
- What happens if the treatment works for me?
Being informed helps you make the best decision for your care.
Talk to a Nurse About Mesothelioma Clinical Trials
Finding the right mesothelioma clinical trial can be time-consuming and confusing. Our experienced nurse can:
- Identify active Phase 2/3 and Phase 3 trials
- Match you based on your diagnosis
- Help coordinate appointments with leading cancer centers
- Answer questions about eligibility and next steps
Contact us today to speak with a nurse and get connected to mesothelioma clinical trials near you.
About Mary Hesdorffer
- Former Executive Director, Mesothelioma Applied Research Foundation (curemeso.org)
- Contributor to research with the National Cancer Institute
- Co-author with leading mesothelioma specialist Dr. Harvey Pass
Read Mary Hesdorffer’s full bio here.
Why getting help from an expert matters
- Avoid delays in care
- Identify the right doctors faster
- Understand complex treatment decisions
- Access clinical trials you may not otherwise find
Frequently Asked Questions (FAQ)
Is there a cost to speak with Mary?
No. This is a free service provided to help you understand your options.
Will this affect my legal case?
No. This is strictly medical guidance and support.
How quickly will I hear back?
Typically, within one business day.